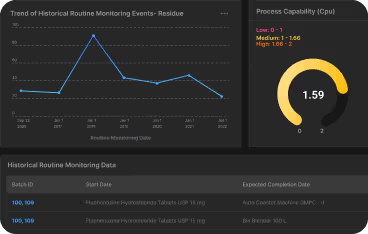
Limits for Multi-API Products

This article delves into the calculation of cleaning limits for products containing multiple Active Pharmaceutical Ingredients (APIs), an essential component of Cleaning Validation processes. Professionals in the field are expected to be familiar with the concepts of Cleaning Limits; for a foundational understanding, refer to "A Discourse on Residue Limits".

Getting Started
When dealing with pharmaceutical products, particularly those with multiple APIs, understanding their individual and combined impacts is crucial. Consider two hypothetical products for illustration:
- Product A: A multi-API tablet containing two APIs, a1 (300 mg, PDE 5 mg, Max DD 900 mg) and a2 (250 mg, PDE 10 mg, Max DD 500 mg), with a total weight of 750 mg.
- Product B: A single-API tablet containing API b (500 mg, PDE 1 mg, Max DD 1000 mg), with a total weight of 650 mg.
Through this framework, we can discuss the concept of "Worst Case APIs" and proceed to elaborate on specific case studies.
Get step-by-step guidance to master API calculation formulas that align with FDA and ICH expectations and on calculating safe carryover limits with precision and compliance in mind.
Case Studies for Multi-API Products
Case 1: Multi-API is Previous Product
In scenarios where Product A (multi-API) precedes Product B (single-API) in manufacturing, we must address the potential carryover of APIs a1 and a2 into Product B. The Acceptable Residue Levels (ARL) for each API in Product A towards Product B are calculated using the formula:

Here, LDDB represents the Largest Daily Dose of Product B, calculated based on its maximum daily dose and tablet weight. Employing this formula yields ARL values for a1 and a2, guiding the establishment of validation protocols.
Case 2: Multi-API is Subsequent Product
Conversely, if Product B is manufactured before Product A, we explore the carryover of API b into Product A. This situation requires a different approach due to the presence of multiple APIs in Product A. By comparing the Largest Daily Doses (LDD) calculated for each API individually, we determine the most restrictive limit to ensure safety.

Validation Protocols and Limit Calculations
Two main approaches emerge from our case studies:
- Dual API Analysis: Sample and test both APIs from Product A, employing their individual limits in the validation protocol.
- Worst API Concept: Focus solely on the API deemed 'worst' based on predetermined criteria (e.g., solubility, toxicity), streamlining the validation process while ensuring safety.
Through detailed examples provided in the discussion, professionals can apply these methodologies to their specific contexts, ensuring comprehensive and compliant cleaning validation.
Streamline validation and scale compliance with Leucine —built for fast-moving pharma teams.
Frequently Asked Questions